
Research and Relationships Define 20 Years of Rotavirus Vaccines
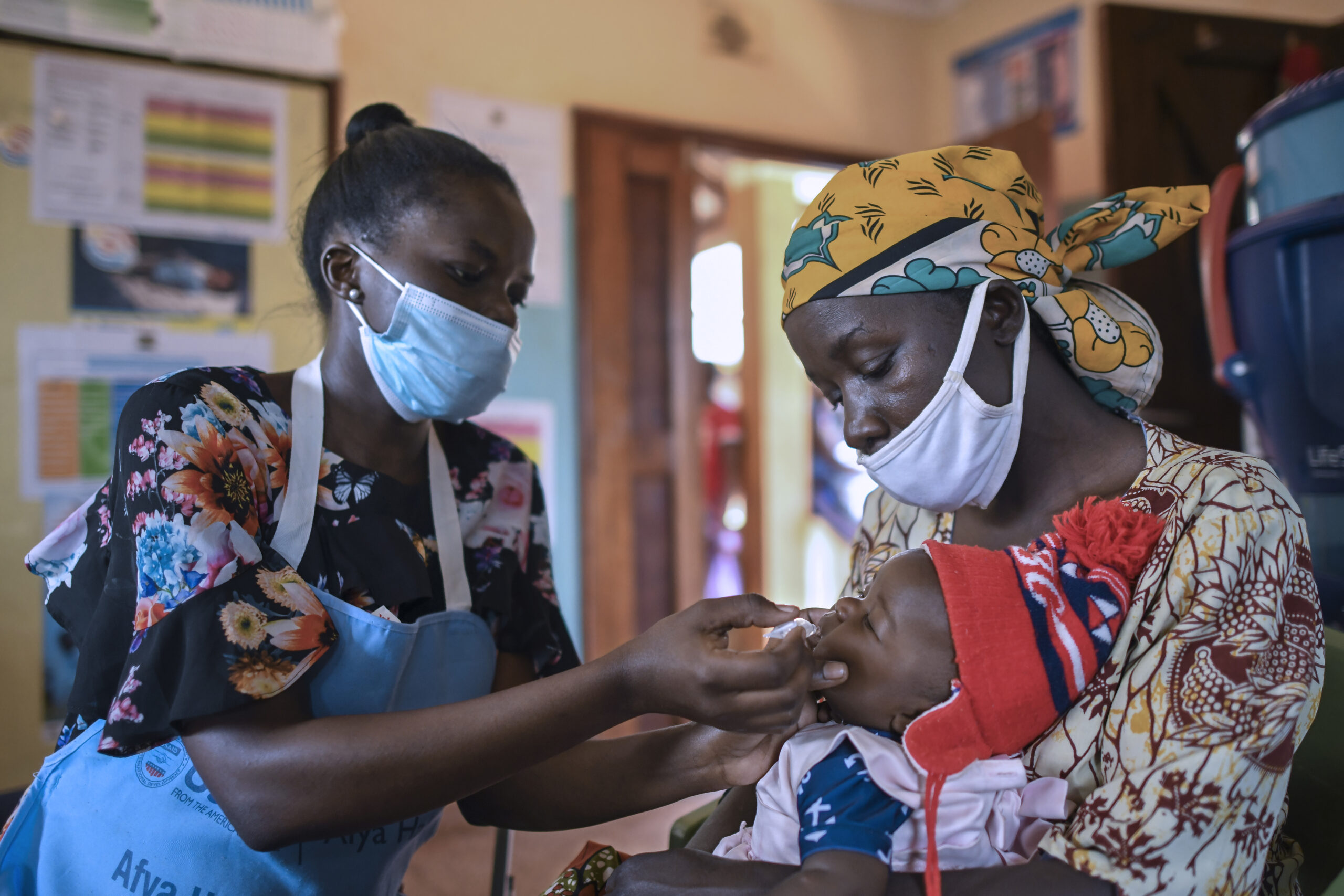
Annet Onyamasi administers an oral rotavirus vaccine to a child at the Khwisero Health Clinic in Kenya. Photo: PATH.
The world has quickly become accustomed to vaccines on demand, as scientists and pharmaceutical companies pursue and release the latest formulations for next-generation COVID boosters. This pace of scientific advancement is exhilarating, at a scale unimagined even 20 years ago.
That’s how long PATH has been pursuing rotavirus vaccines, to protect the world’s most vulnerable children from this “democratic disease.” Throughout our long-term investment, PATH has served as a scientific expert, bridge, and convener (see timeline below). Today, rotavirus vaccines are a global success. But our work is not over.
Back in 2003, rotavirus killed half a million children every year, mostly in low-income communities of sub-Saharan Africa and Southeast Asia. It attacks the gut and causes severe diarrhea, vomiting, and dehydration. In wealthy countries, rotavirus is primarily an inconvenience. Treatment is available at any grocery store. But in rural, high-burden communities, these products can be difficult to access. Sick babies dehydrate quickly, and homemade treatment relies not only on supplies but clean water. When dehydration becomes severe—and clinical care is out of reach—rotavirus can be fatal.
Twenty years ago, Merck and GSK were developing rotavirus vaccines. But a 15 to 20-year gap was typical between new vaccine introduction in high-income and low-income countries, respectively. Clinical trials had only studied the vaccines’ performance in the US, Europe, and Latin America. No one knew if they would protect children where they were needed the most. Gavi, the Vaccine Alliance had rotavirus vaccines on its radar, but their funding support to the most vulnerable countries was contingent on evidence that did not yet exist. That’s when Gavi funded PATH and other partners to generate this evidence.
“PATH partnered with the pharmaceutical companies and the World Health Organization to sync development of rotavirus vaccines for markets in high- and low-income countries,” explained Dr. Deborah Atherly, Global Head of Policy, Access, and Introduction; PATH Center for Vaccine Innovation and Access. “At that time, public-private partnerships in the vaccine space were not common.”
PATH initiated clinical trials of the Merck and GSK vaccines in Ghana, Kenya, Malawi, Mali, South Africa and Bangladesh. We tapped regional networks for medical expertise and built local research capacity to ensure that the studies were conducted at the highest standards. PATH welcomed national ministries to the conversation, for strategies that leveraged local context. “Thoughtful engagement is critical,” Dr. Atherly emphasized.
PATH also conducted studies on vaccine economics, delivery strategies, and local programmatic considerations. By 2009, the vaccines were proven safe and efficacious worldwide and achieved WHO approval and Gavi financing. The evidence base that PATH and our partners had built was essential for informing country applications for the new vaccines.
In 2011, Sudan became the first Gavi-eligible country to introduce rotavirus vaccines in the public sector. Introduction in the United States had come five years earlier, and Nicaragua broke records by marking this achievement that same year, too. The gap had closed between high- and low-income access to this new vaccine. Over the next five years, rotavirus vaccines reached across 70% of the African continent.
“Vaccination is a children’s right,” remarked UNICEF representative Félicité Tchibindat at the 2014 rotavirus vaccine launch in Cameroon, “It is important for parents to get their children vaccinated with the rotavirus vaccine… to ensure the survival and full growth of children.”
With global demand growing, vaccine options and supply would need to grow, too. PATH anticipated this evolution, building early partnerships with manufacturers in India and China to study new vaccine candidates that could be produced at lower cost. The Bill & Melinda Gates Foundation funded PATH’s long-term collaborations with Indian manufacturers Bharat Biotech and Serum Institute of India leading to two additional rotavirus vaccines that have also proven their safety and efficacy. In 2018, these were recommended for global use by the World Health Organization.
By January 2023, 123 countries had introduced rotavirus vaccines into their public sector health programs. The gains are dramatic, and rotavirus vaccines have saved thousands of lives. But rotavirus still kills 128,000 children every year, and eight countries in sub-Saharan Africa carry 80% of the global burden. Nigeria, the country carrying the highest global burden, just gave the global fight a vital boost, introducing the Bharat Biotech rotavirus vaccine into its routine immunization program in late 2022.
Rotavirus vaccines have made halting progress in Southeast Asia, where economic disparities within countries extend to the health sector. High rotavirus burden occurs in sub-national regions of many Asian countries, and alternative introduction strategies may be among the key points of discussion at the upcoming International Rotavirus Symposium, held this year in Bali, Indonesia. “The symposium is a forum to talk with country representatives and hear their perspectives on these challenges,” said Dr. Atherly.
Creative approaches and collaborations have been a hallmark of the 20-year path to global access for rotavirus vaccines. The work is not over, and PATH’s partnerships in research, advocacy, and technical assistance sustain. New candidates like injectable rotavirus vaccines or alternate formulations could improve efficacy and increase supply options for the highest-burden settings. In countries where they have been introduced, coverage with rotavirus vaccines also has significant room for improvement.
For vaccines to achieve their greatest potential, it requires engaging, equipping, and motivating a complex network of partners, and funded efforts to continue to optimize rotavirus vaccine performance, supply, and coverage. The past 20 years marked an auspicious beginning, followed by lifesaving milestones across the world. The fight against rotavirus carries on, and PATH will continue to champion partnerships in scientific research, clinical development, manufacturing, economics, advocacy, and policy that ensure all children are protected.
PATH Rotavirus Experience
Milestone (icon) categories of PATH efforts
- Economics (E)
- Advocacy (A)
- Research (R)
- Technical Assistance (T)
Timeline of Key Dates
2001: PATH partnerships in India and China advance early candidate rotavirus vaccines (R, T)
2003: Gavi, the Vaccine Alliance funds PATH’s Rotavirus Vaccine Program to accelerate access in high-burden, low-income countries (E, A, R, T)
2006: PATH partnerships in China advance early candidate vaccines. (R, T)
2006: WHO recommends available (US- & Europe-manufactured) rotavirus vaccines for the Americas and Europe. (R, A)
2006: United States & Nicaragua introduce rotavirus vaccines, marking the first time a vaccine is introduced in high- and low-income countries in the same year; PATH helped facilitate Nicaragua’s access to the vaccines. (R, A)
2007: Based on the investment case developed by PATH, WHO, and CDC, Gavi approves rotavirus vaccine funding for Latin America and Europe.
2007: PATH begins Phase 3 clinical trials to test available rotavirus vaccines in high-burden communities of Africa and Asia. (R, T)
2009: WHO issues global recommendation for available rotavirus vaccines (A)
2009: High-burden countries apply to Gavi for rotavirus vaccine financing. (E, A)
2011: Sudan becomes the first in a cascade of several African Gavi-eligible countries to introduce rotavirus vaccine in its national program.
2011: Phase 3 clinical trials begin for first India-manufactured vaccine candidate (R, T)
2014: Phase 3 clinical trials begin for second India-manufactured vaccine candidate. (R, T)
2011: PATH studies next-generation, injectable rotavirus vaccine candidates (R)
2014: Government of India approves the first locally manufactured rotavirus vaccine for public-sector introduction. (R, A)
2016: India’s national immunization program introduces rotavirus vaccines (A, E)
2017: Government of India approves second locally manufactured rotavirus vaccine for public sector. (R, A)
2018: WHO approves India-manufactured rotavirus vaccines for procurement by UN and Gavi, a critical step for low-income country access. (R, A)
2019 Phase 3 clinical trials begin for injectable rotavirus vaccine candidate.
2019-2020 PATH conducts bridging study for rotavirus vaccine manufactured in Vietnam. (R)
2019-2021 PATH studies optimized formulation of India-manufactured rotavirus vaccine in Zambia. (R)
2020: PATH develops rotavirus vaccine cost-calculator (E, A)
2020-2021: PATH conducts studies on cost-effectiveness, feasibility, and acceptability of injectable rotavirus vaccine candidates (E, R)
2021: WHO global recommendation includes India-manufactured rotavirus vaccines (R, A)
2021-2022: PATH helps countries evaluate new vaccine options and strategies (E, R)
2023: As of January 2023, 123 countries provide rotavirus vaccines in public-sector immunization programs (E, A, R, T)


