
Health benefits of rotavirus vaccination in Palestine continue through a product switch
Rotavirus, the leading cause of severe childhood diarrheal illness and deaths, represented a significant public health burden in Palestine prior to the introduction of rotavirus vaccination. Surveillance of diarrheal and rotavirus-specific disease has documented the substantial health impact of rotavirus vaccination in many countries, and Palestine is no exception.
We recently partnered with the Palestinian Ministry of Health (MOH) to conduct a vaccine impact analysis examining the health benefits of rotavirus immunization for children in Palestine. The results confirm that rotavirus vaccination has significantly reduced the burden of diarrhea among children in Palestine. Furthermore, the benefits were sustained through a vaccine product switch.
Rotavirus vaccination in Palestine
In 2016, Palestine added the rotavirus vaccine ROTARIX® to its routine immunization program. One of our organizations, the Rostropovich-Vishnevskaya Foundation (RVF), worked with the Palestinian MOH to demonstrate the health impact of the rotavirus vaccination program in children by analyzing surveillance data prior to and for two years following the introduction of ROTARIX. The results indicated a 27 percent drop in outpatient diarrhea cases in Gaza. In the West Bank, surveillance at a large children’s hospital showed a 65 percent decrease in rotavirus prevalence among pediatric outpatient and inpatient diarrhea cases.
In 2018, the MOH decided to switch to ROTAVAC®, a more affordable rotavirus vaccine made in India that had recently been prequalified by the World Health Organization (WHO). Both of our organizations, RVF and PATH, collaborated with the MOH on an economic analysis which confirmed that, while either vaccine was cost-effective, the switch to ROTAVAC was cost-saving. In tandem, and building upon previous surveillance work, we also worked together to track health indicators for two years following the switch to ROTAVAC in order to confirm the continued health benefits of the rotavirus immunization program.
Results of the impact analysis
Between 2016 and 2020, rotavirus vaccination resulted in a 54 percent decrease in diarrhea overall in Gaza and a 69 percent decrease in rotavirus positivity rates in the West Bank. These data indicate a continued decline in diarrheal cases while using ROTAVAC, confirming that the benefits of rotavirus vaccination have been sustained through a product switch.
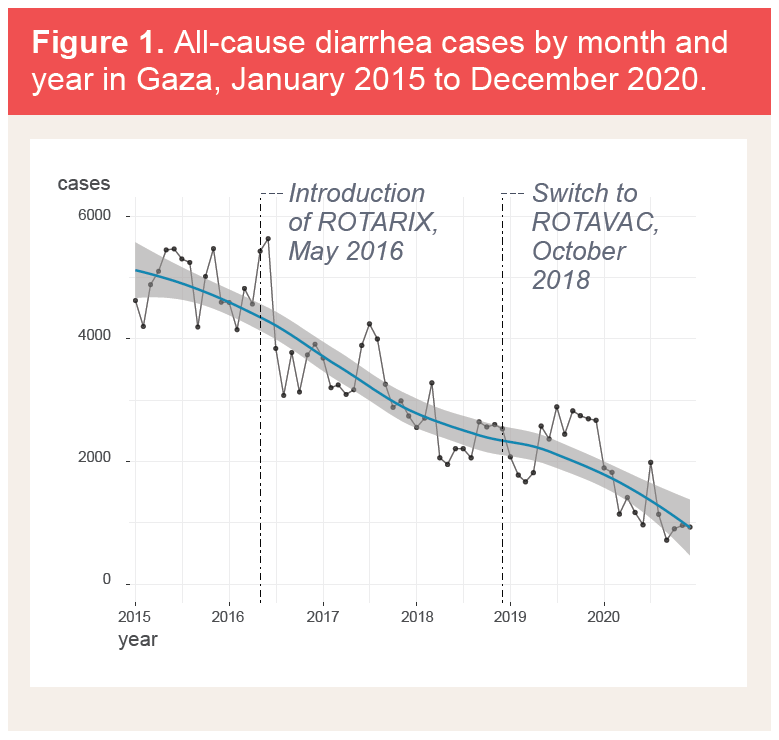 Diarrhea cases in Gaza dropped after the introduction of ROTARIX and continued to decline after the switch to ROTAVAC.This impact analysis encompassed two different surveillance activities, one tracking clinical diarrhea cases of any cause, and the other tracking rotavirus-specific diarrheal disease. Despite these differences, these two sets of data provide complementary indications of rotavirus vaccine impact in Palestine. One provides scale, and the other provides specificity. Both assessments present strong evidence that the rotavirus immunization program has had a substantial impact on reducing the burden of rotavirus disease among children in Palestine.
Diarrhea cases in Gaza dropped after the introduction of ROTARIX and continued to decline after the switch to ROTAVAC.This impact analysis encompassed two different surveillance activities, one tracking clinical diarrhea cases of any cause, and the other tracking rotavirus-specific diarrheal disease. Despite these differences, these two sets of data provide complementary indications of rotavirus vaccine impact in Palestine. One provides scale, and the other provides specificity. Both assessments present strong evidence that the rotavirus immunization program has had a substantial impact on reducing the burden of rotavirus disease among children in Palestine.
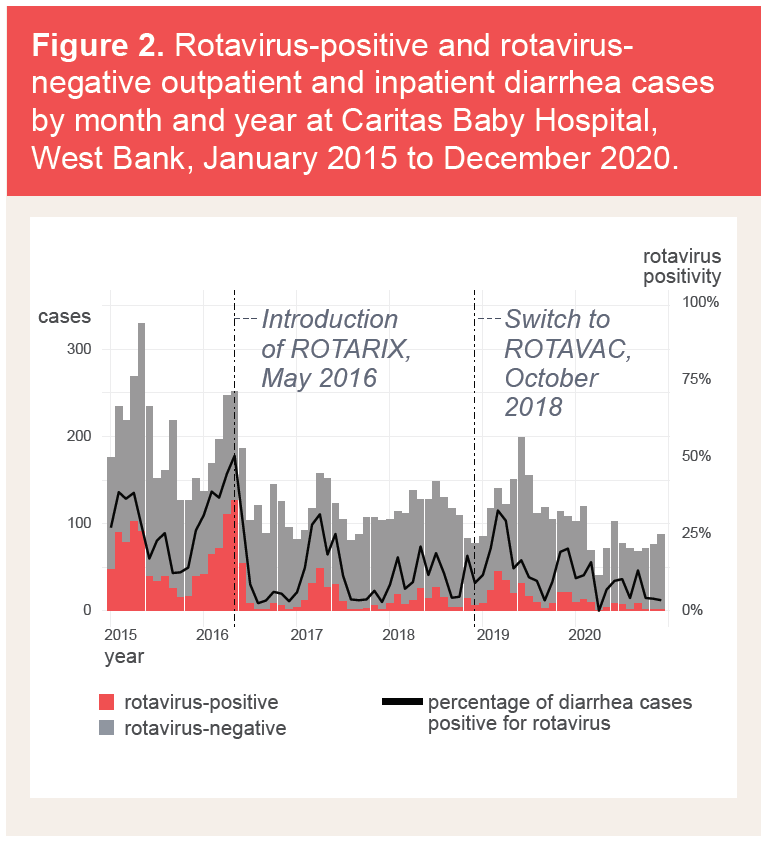 Rotavirus-specific surveillance data in West Bank showed a sharp decline in rotavirus positivity after rotavirus vaccine introduction that was maintained after the product switch.Some of these data were gathered during the COVID-19 pandemic. While disruptions appear to have been minimal, work is ongoing to fully understand the impact of pandemic-related disruptions on health care utilization in both Gaza and the West Bank.
Rotavirus-specific surveillance data in West Bank showed a sharp decline in rotavirus positivity after rotavirus vaccine introduction that was maintained after the product switch.Some of these data were gathered during the COVID-19 pandemic. While disruptions appear to have been minimal, work is ongoing to fully understand the impact of pandemic-related disruptions on health care utilization in both Gaza and the West Bank.
Implications for Palestine and beyond
We hope that these results demonstrate to other countries that a rotavirus vaccine product switch is possible, and that the reduction of diarrheal illness and deaths among young children can be sustained through the switch process without interruption. Any WHO-prequalified rotavirus vaccine can successfully reduce the burden of rotavirus diarrhea in young children. Assessing a variety of characteristics, including cost, cold chain requirements, and doses required, will help decision-makers choose the best vaccine for their country.
Children in Palestine and all over the world where diarrheal disease burden is high face many challenges. It’s reassuring to now know with certainty that suffering from rotavirus still does not need to be one of them.


