
Made in Bangladesh, this rapid diagnostic test could be the key for early detection of cholera
Cholera is a major health burden in low- and middle-income countries. Globally, an estimated 1.3 billion people are at risk. South Asia constitutes the largest share of this at-risk population group, including at least 66 million in Bangladesh, where over 100,000 cases are reported annually. Cholera is an infectious disease characterised by severe watery diarrhoea and vomiting. Left untreated, the rapid dehydration it causes can be deadly.
In the 1960s, researchers at icddr,b and its partners developed a simple solution to combat cholera – oral rehydration therapy (ORT) – a mixture of salt, sugar, and water, ingredients readily found in most homes. It has saved nearly 50 million lives from diarrhoeal diseases. However, cholera is often misdiagnosed until the patient is in an extreme condition, when ORT is not enough to prevent death by dehydration. Thus, early detection is critical in saving lives and preventing further spread of the disease.
Diagnosis for cholera needs to be quick, easy, and accurate. Stool cultures have until now been the specimen of choice for diagnosis. But Rapid Diagnostic Tests (RDTs) are gaining momentum because they yield results in a matter of hours – crucial in a life-or-death situation.
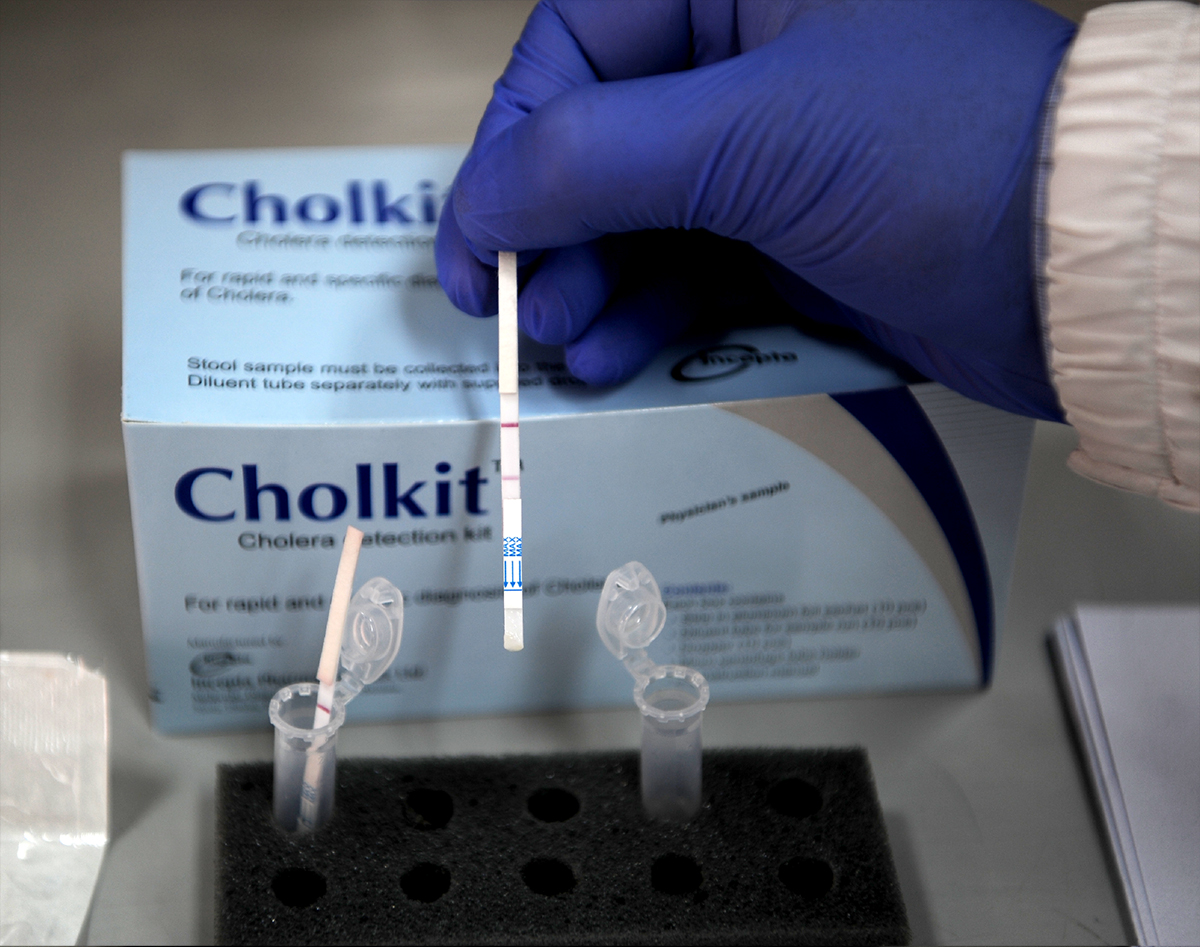
The serogroups O1 and O139 of the bacteria Vibrio cholera are to blame for cholera pandemics and endemics. One of the most used RDTs currently available is Crystal VC, a dipstick assay which detects both O1 and O139 serogroups. While Crystal VC has shown promising results in sensitivity for detection, the presence of both serogroups can lead to lower specificity. To counter this deficiency, scientists at icddr,b, in collaboration with Incepta Pharmaceuticals, have developed a cholera RDT – CholKit – which is based on the O1 serogroup only, as there have been no epidemics reported due to the O139 serogroup in the past decade. It is the first and only cholera RDT to be developed in Bangladesh.
A recent study compared the performance of the two RDTs, CholKit and Crystal VC. It was performed first at icddr,b’s Dhaka hospital and then expanded to 22 sentinel cholera surveillance sites across Bangladesh. CholKit performed with similar sensitivity and specificity as the currently available Crystal VC. However, the detection of only V. cholerae O1 is advantageous because it avoids false positive test results of V. cholerae O139 that can happen with Crystal VC.
CholKit has received licensure in Bangladesh and is awaiting mass production to make it available in the market place. The innovation itself, the first of its kind to test V. cholerae O1, was designed to be replicated in other resource-poor countries similar to Bangladesh where there are persistent threats of a cholera outbreak. It is now being used in other cholera-prone countries to test its performance in similar settings.


