
'Super Bif’ to the rescue! How the infant gut microbiome helps fight infections
The gut microbiome is an area of great interest for researchers and clinicians alike. While we know that the bacteria, viruses, and parasites that live in our gut have a huge impact on our health, we are still learning exactly which ones are most beneficial, how they interact with each other, and and how they affect our health and susceptibility to infections. For Prof. Lindsay Hall, a microbiologist at the Technical University of Munich who focuses on the early infant gut microbiome, one of the most important—and fascinating—species of gut bacteria is called Bifidobacterium (or Bif, for short). Her research has helped explain how Bif actually fights infections such as diarrheal disease—and how supplementation could save lives. I spoke with Dr. Hall to learn more.
 Professor Lindsay Hall is a microbiologist at the Technical University of Munich who focuses on the early infant gut microbiome. Learn more about her lab.Laura: Why does the gut microbiome matter for a child’s health and well-being?
Professor Lindsay Hall is a microbiologist at the Technical University of Munich who focuses on the early infant gut microbiome. Learn more about her lab.Laura: Why does the gut microbiome matter for a child’s health and well-being?
Prof. Hall: We’re not just vessels for our microbes. These microbes do a huge range of functions for us, and this starts at birth when these microbes are basically transferred from mum to baby. They help program your immune system. They help digest the food that we eat, and they’re really important when it comes to infection resistance. And there’s good evidence to indicate that they’re important for brain development as well. They do a lot of different things for us, which is why having a healthy, balanced gut microbiota when we’re infants is really important. It basically programs nearly all aspects of our physiology.
Laura: How was Bifidobacterium first identified as such an important probiotic for young infants?
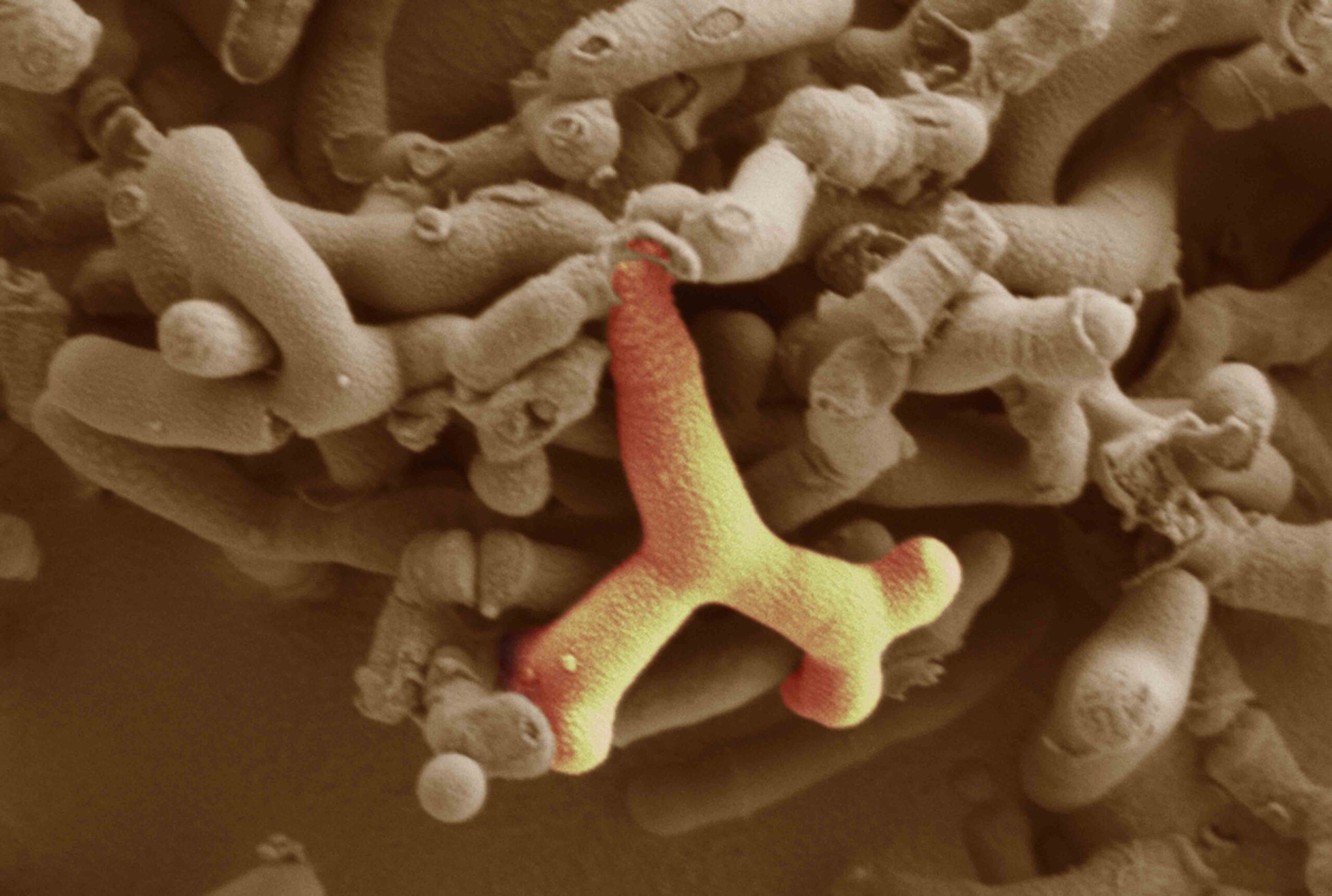
Prof. Hall: Even if you don’t work in the field at all, just look at the supermarket shelves and you can normally see Bif promoted as being good for you. This tradition comes from the Pasteur Institute, back in 1899. A French pediatrician, Henry Tisser, had a cohort of babies with diarrhea and other babies that were very healthy. And when he looked at the poo samples of the healthy babies, he noticed these Y-shaped bacteria that were missing in the babies that had the diarrhea. When he isolated these Y-shaped microbes and gave them to the babies with diarrhea, they got better. This was Bif. Then everybody started realizing that not all microbes are bad for us; in fact, the majority are actually very helpful.
Laura: How and why is Bifidobacterium so beneficial?
Prof. Hall: One reason is that Bif can utilize sugars from breast milk that other microbes cannot, so they have a selective advantage. Breast milk essentially feeds Bif. They can then produce these metabolites that strengthen the immune system. Additionally, the metabolites produced by Bif are acidic, so they reduce the gut’s pH. A lot of harmful microbes don’t like low pH, so this basically inhibits their growth. Some types of Bif may also produce novel antimicrobial compounds that act like antibiotics, directly inhibiting disease-causing microbes. We call these “Super Bif” because they’ve got so many beneficial aspects.
Laura: At the VASE conference, you talked about a study you recently completed looking at Bif supplementations in young infants that had promising results. Could you tell me about that study?
Prof. Hall: I presented some of our findings from our BAMBI preterm baby cohort in the UK. Many preterm babies are born via C-section, which disrupts the transfer of beneficial microbes from mum to baby, and are often given antibiotics, which can kill the good bacteria with the bad. This makes preterm babies especially vulnerable to microbiome disturbances and, as a result, infections. So we wanted to see if supplementing with Bif would help.
We had one group of preterm infants that was routinely supplemented with our probiotic containing Bifidobacterium bifidum, and we had another group that was not supplemented—just routine clinical care. We took poo samples from the babies the whole time they were in the neonatal intensive care unit and watched to see if that changed the developing microbial ecosystem over time.
We found that the supplemented babies had high levels of Bif even just one week post birth. We didn’t see this in our control babies; we did see Bif coming in, but it was much later. We also saw a concurrent reduction in potentially dangerous bacteria like Enterococcus, Klebsiella, or Staphylococcus—microbes we don’t want in the babies’ gut—along with a complete change in the gut environment based on the beneficial compounds that Bif produces. Then, working with our clinical colleagues, we showed that routine supplementations resulted in an over 50% reduction in two devastating diseases in preterm babies—late onset sepsis and necrotizing enterocolitis—that are associated with infection and overgrowth of disease-causing microbes. So, there was a really positive clinical outcome as well.
Laura: That’s incredible. What are some of the potential applications for this research?
Prof. Hall: If you have a preterm baby, I think giving a Bif supplement is a good thing. And I think we need to think carefully about choosing the right Bif strain. We may also need to think even earlier and consider nutrition for mum. Can we supplement mum with Bif? She may be lacking it, and if she is, she’s never going to pass it on to baby. If mum’s not healthy then that’s going to negatively impact baby, no matter how much we supplement.
It might also be that, depending on where your babies are in the world, you might need a different Bif strain that may be effective against different pathogens. That’s the study that we’re just getting started.
The other thing we’re beginning to look at is actually boosting immune response to vaccination. We know that some babies don’t respond the same to different types of vaccines as others, so you could give Bif supplements to give an additional boost to get better anti-pathogen responses.
I think these are very realistic possibilities—and really cost effective, too.
Laura: This is such a promising and exciting area of research. We’re excited to see what you do next.
Prof. Hall: Thanks. Yeah, well, there’s so much to do!


